What’s new in Opcenter Research Development and Laboratory 2204

Gain enhanced usability, multidimensional scheduling and extended cloud capabilities
We are excited to announce the availability of Opcenter™ Research Development and Laboratory software version 2204! Version 2204 stands for the April (04) release of the year 2022. 2204 is the 9.2 planned release and the next release will be 2210 (in October). The new versioning (YYMM) will be used across all manufacturing operations management (MOM) products. 2204 is a major Opcenter RD&L version like others in the past (9.0, 9.1).
Integration with Opcenter APS Testing and verification Opcenter RD&L provides great value when it comes to organizing the execution of tests on prototypes during research and development (R&D). Companies must optimize the use of multiple test facilities and expensive equipment. By having a bidirectional integration, Opcenter RD&L version 2204 and Opcenter APS 2204 work seamlessly together in assigning and scheduling different tests to be executed.
Opcenter RD&L 2204 brings you the following benefits:
- Gain enhanced usability, seamless integration with Opcenter APS for scheduling and extended cloud capabilities with Xcelerator Share
- Rapidly exchange product data with partners
- Reduce administrative tasks and errors
- Maintain product quality and compliance
- Reduce costs in new product development and introduction
- Standardize intellectual property throughout the company
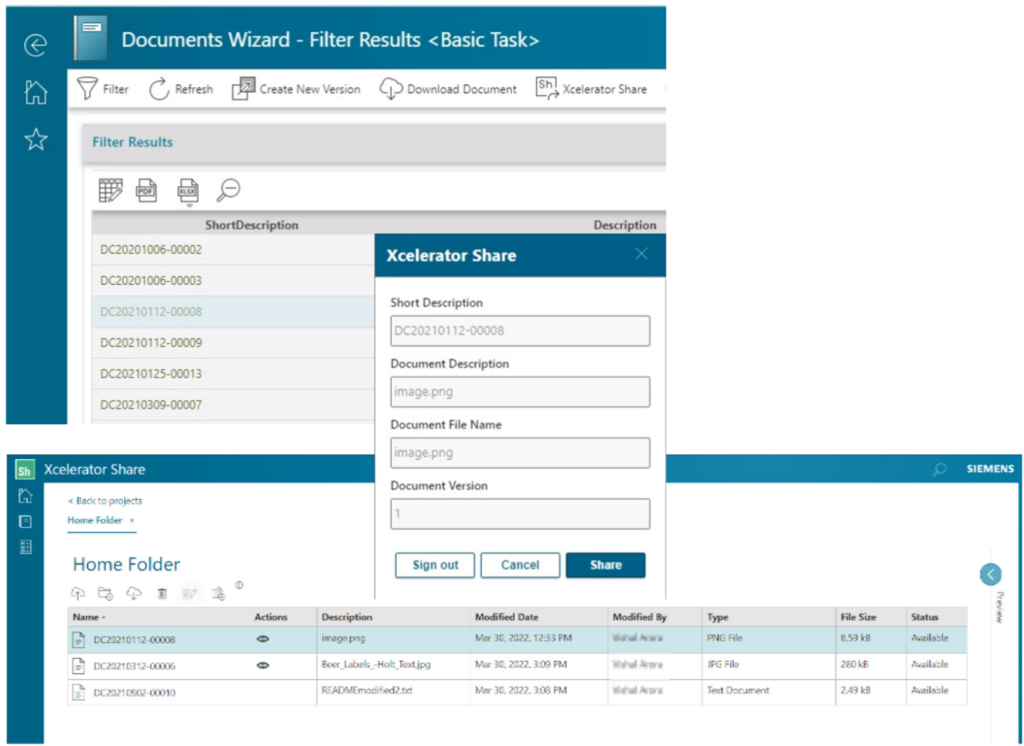
Integration with Xcelerator Share
Xcelerator Share is a cloud-based sharing platform offered by Siemens, which provides greater flexibility for our customers. By integrating with Xcelerator Share, Opcenter RD&L users have the option to share documents uploaded in RD&L with other (not necessarily RD&L) users.
Already using Opcenter RD&L? See what’s new in version 2204.
Want to learn more? Opcenter RD&L offers companies in CPG and process a scalable and flexible platform to streamline, optimize, and align all formulated product data management. That way they can capitalize on their R&D potential and develop new products faster. Opcenter RD&L keeps the product designs and processes well in-line with quality and regulatory requirements. The smooth integration and alignment of R&D and manufacturing data and processes drastically speeds up the transfer of final product designs to mainstream manufacturing.
Typical activities such as project data management, formula development, trial and experiment management, and production process design require an integrated set of best-in-class components ranging from electronic notebooks to laboratory management over specification management to supplier collaboration, pilot plant manufacturing and the like. Key functionalities like regulatory compliance and support for the Intellectual Property process are an integral part of Opcenter RD&L. Find out more about Opcenter RD&L


