How manufacturing digitalization can help MD&D companies: On-demand webinar
With digitalization becoming a pervasive change agent in nearly every aspect of our lives – from ebooks apps for pharmaceutical prescriptions and fulfillment – it is no surprise that digitalization is impacting much of what MD&D manufacturers do today. Changes tend to be customer-driven, with patient expectations migrating to more active and collaborative patient-provider relationships and demanding information about diagnoses and treatment choices as well as cost transparency.
Technological innovation for medtech products also relies more and more on digitalization: connected diagnostics, customized orthopedics, next-generation implants, needle-free diabetic care, and the Internet of Medical Things (IoMT), to name a few examples. Just as these new product introductions have been made possible by digitalization, so too digitalization is transforming the way MD&D manufacturers approach their businesses.
Through digitalization, medtech companies have access to a wealth of data from product design, process engineering, and manufacturing sources, as well as product utilization by the patients themselves. Data analytics enable rapid response to the information and insights gleaned from these data sources.
As the MD&D industry navigates increasing complexity in products, globalization of supply chains, rapid changes in regulatory landscapes, and implications of the IoMT, it needs digital technologies that empower every phase of the innovation process, from ideation to realization and utilization – integrated via a digital thread for needed connectivity. Realizing the full value of digitalization through the digital enterprise represents both a great challenge and a great business opportunity for medtech companies – one that Siemens is able to help your company tackle.
View the on-demand webinar to understand the impact manufacturing digitalization is having on MD&D manufacturers and to learn more about Siemens’ proven approach to integrated manufacturing execution systems (MES), including:
- Benefits of MD&D digitalization
- The key roles of MES and manufacturing operations management (MOM), including:
- The integration of technical domains into one data model
- The collaboration platform, involving digital twins
- Orchestration of manufacturing and quality operations
- Data contextualization with manufacturing information
- MES and other MOM elements, and how each benefits the manufacturer
- Meeting FDA expectations in the agency’s shift from regulatory compliance to quality management
- Enterprise strategies for implementing effective digitalization
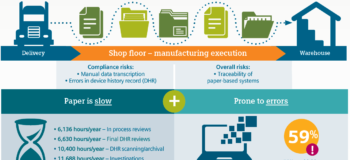
Download the the infographic here to get insight !


