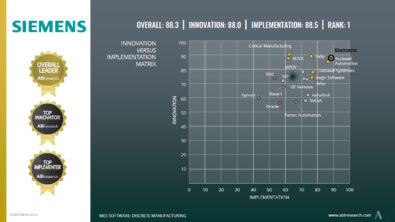
Good news for MedTech manufacturers: Siemens MES positioned as Leader by Gartner
Siemens has been named by Gartner, Inc. as a “Leader” in its 2017 Magic Quadrant for Manufacturing Execution Systems/Manufacturing Operations Management (MES/MOM).
Why is that good news for Medical Device Manufacturers?
Because our MES/MOM for the Medical Device industry is built on the leading technology in the world. As you look to invest in technology that is going to take you into the future, you can be confident that when you select Siemens as your partner, you have the best opportunity for success.
Gartner is a leading worldwide IT research and advisory firm. Each year, Gartner evaluates vendors and displays rankings in the Magic Quadrant. In the 2017 Gartner MES/MOM Magic Quadrant, which is the first ever for this area of applications, companies were assessed on “Ability to Execute”, covering aspects such as products or service, overall viability, market responsiveness/record, marketing execution, operations and customer experience along with “Completeness of Vision”, which scores aspects such as product strategy, innovation, understanding of market and industry verticals.
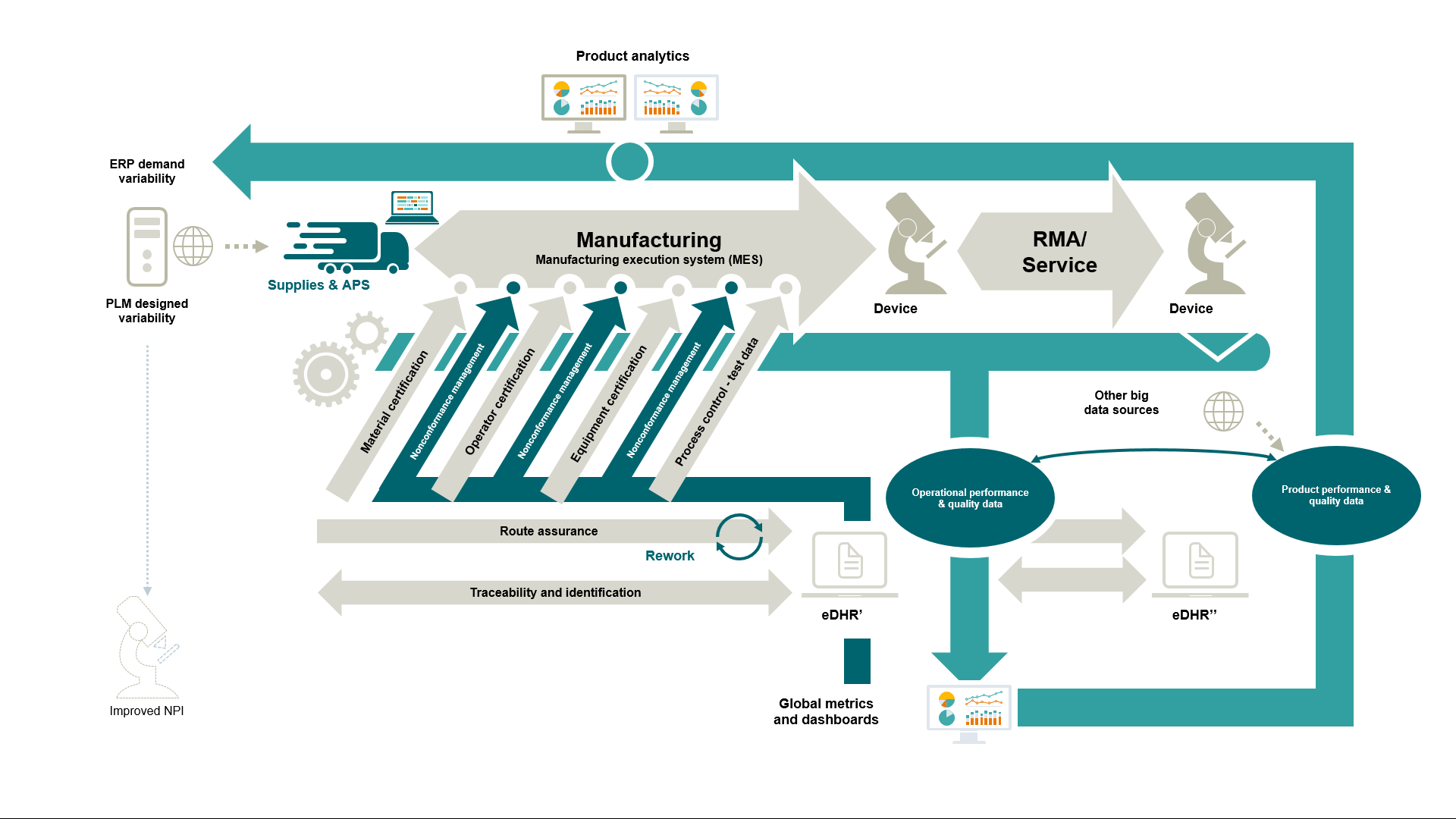
As Siemens we hold a unique position in the manufacturing market by providing a holistic solution that supports the entire value chain, from ideation to design, manufacturing execution, and software for plant automation, plant hardware, coupled with an open cloud-based operating system for the IoT.
For Medical Device manufacturers, this translates to a host of benefits:
- Reduced Cost & Risk of Compliance: automated processes drive tighter, enterprise-wide compliance through an electronic device history hecord (eDHR) or electronic batch record (eBR) while eliminating human errors and oversights. The resulting proven reductions in field product quality issues, complaints, and warranty claims lower product risk and protect your brand while lowered costs strengthen market share.
- Stabilized Supply Chain: upstream and downstream supply chain disruptions are diminished through complete product and process enforcement and traceability. Joined with increased manufacturing velocity, enterprise-wide enforcement, traceability, and visibility generate highly predictable output quality yielding higher customer satisfaction.
- Rapid NPI & Reliable Change Management: design processes are streamlined with concurrent product and process design transfer, reducing new product ramp time, and bringing innovative technologies more rapidly to market. Effective change management is essential to gaining process control and enforcement, significantly reducing variability.
- Improved Profit Margin: revenue growth is accelerated by increasing manufacturing velocity and heightening predictability of product quality to generate larger quantities of final product. Simultaneously, costs are decreased by reducing or eliminating paper, scrap, labor, inventory, and non-value-added activities. The simultaneous effects increasing revenues and reducing costs improve the profit margin at the most rapid rate – meeting or exceeding shareholder/investor expectations.
- Enhanced Decision Making: decision-making capabilities are enhanced by gaining actionable intelligence from dashboards, preconfigured reports, and ad hoc reports correlating granular data from multiple, global, enterprise-wide sources.
- Lean Enablement: lean initiatives are driven through shorter lots, automated poke-a-yoke, paperless manufacturing, end-to-end process control/enforcement/visibility, electronic procedures, operator certification, machine maintenance/calibration enforcement, and integrated electronic lean workflows and kanban.
Have you investigated the leading platform for Medical Device Manufacturing? See what it’s all about
Click here for more information on Siemens’ Magic Quadrant placement, and to view the 2017 Magic Quadrant report for MES/MOM.