Digitalizing medical devices manufacturing
While medical device manufacturers are focused on bringing innovative products to market, many are in recall denial, believing that recalls affect other companies, not them.
Unfortunately, FDA’s reports say otherwise: recalls are woefully on the rise, and pose a huge financial risk to manufacturers.

This reality becomes even more alarming if we consider that Med-Tech companies are managing recalls and non-conformances with paper-based systems. Collecting mounds of data on paper records and PDF reports, retaining and hoarding them to meet statutory and regulatory requirements is synonymous with “being slow, inefficient and prone to errors.” The systems are neither preventing nor effectively containing quality issues.

Is this the right approach towards product quality?
Of course not! Firefighting is a strong indicator that a company is focused on compliance over quality. This reactive approach has had the unintended consequence of creating D.R.I.P. (Data Rich, Intelligence Poor) environments, where dumb documents are managed instead of product quality data for intelligent use.
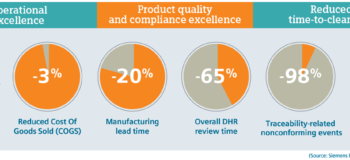
Proactively improving product quality must be the driving principle within all medical device manufacturers.
They must overcome regulatory inertia by transitioning from paper systems to smart Manufacturing Execution Systems. This Digital Transformation will minimize opportunities for error, improve quality and reduce costs, while enabling firms to comply with new regulatory requirements as a natural outcome.




